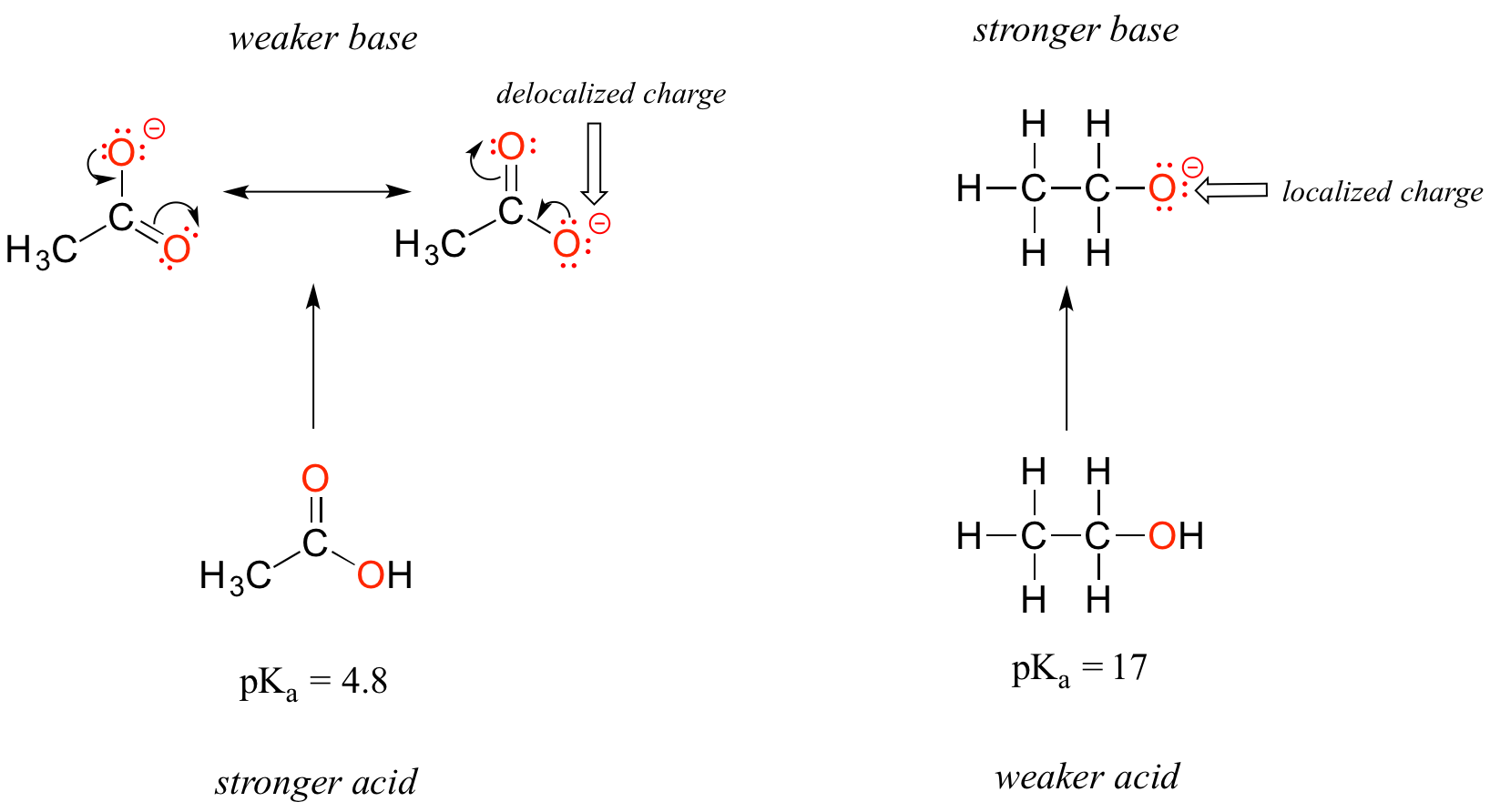
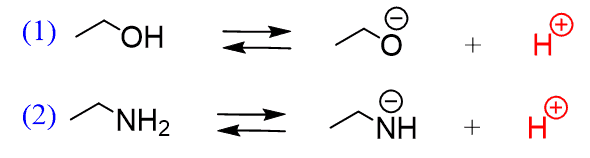
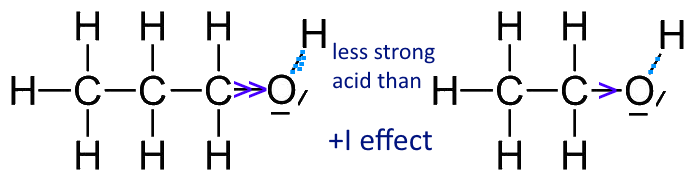
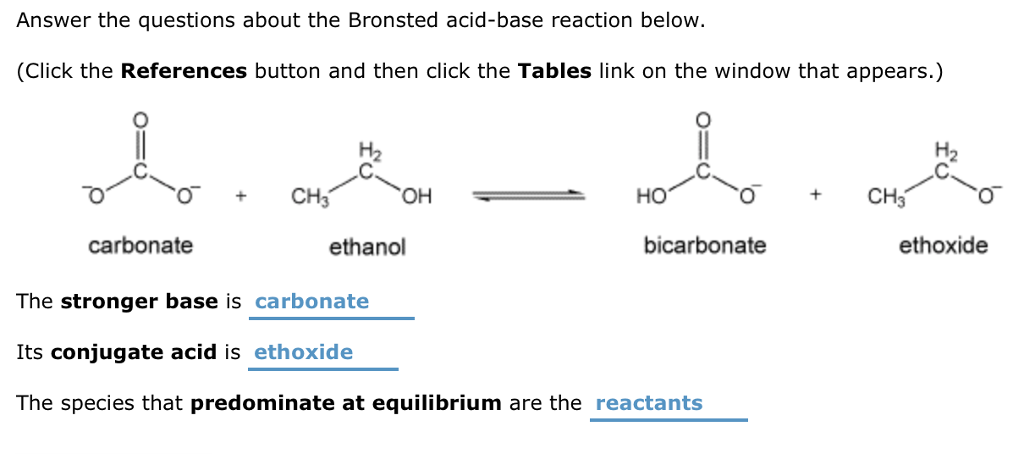
Why is the conjugate acid of an ether/alcohol more acidic than that of a hydronium ion? : r/chemhelp

Mechanism and Kinetics of Isobutene Formation from Ethanol and Acetone over ZnxZryOz | ACS Catalysis

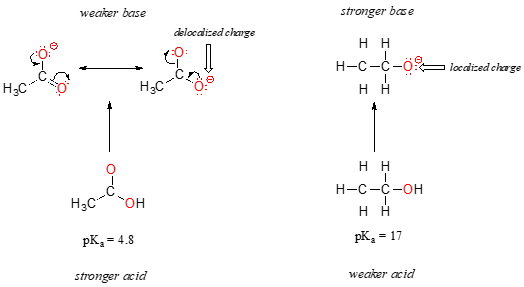
Draw the Lewis structure for the conjugate base from the reaction of ethanol with a generic base. Include all lone pairs of electrons and any nonzero formal charges. | Homework.Study.com
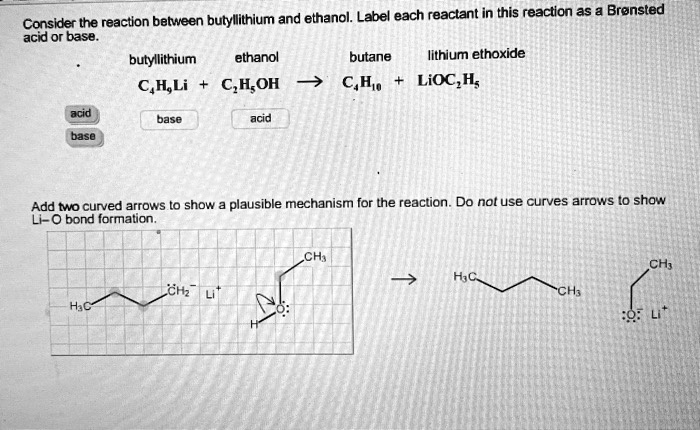
SOLVED: reaction belween butylithium and ethanol: Label each reactant in this reaction as Bronsted Consider the acid or base. butyllithium ethanol butane Ilthium ethoxide CH,Li C,H;OH C,H,o LiOC,Hs acid base acid base
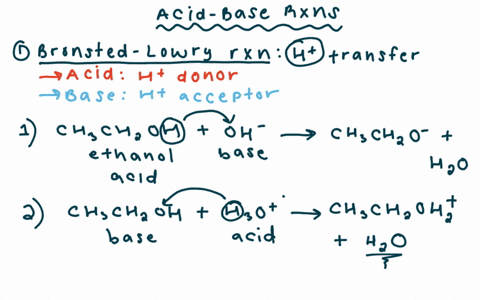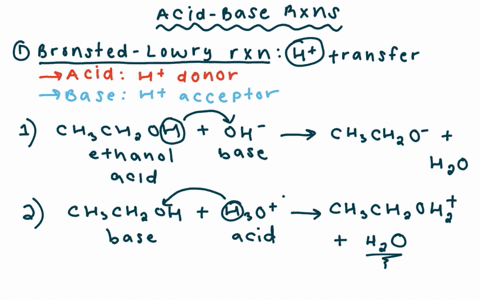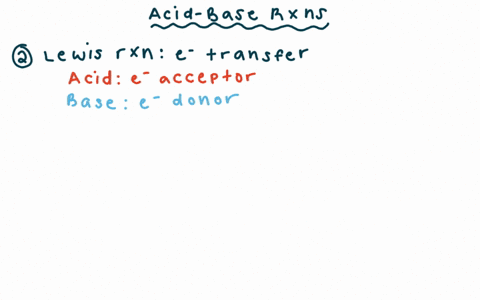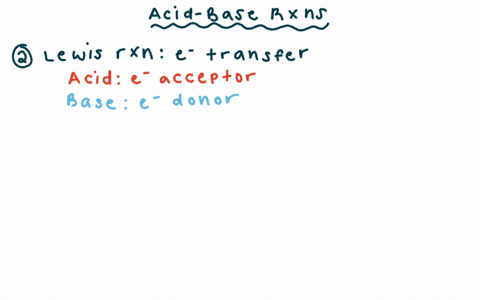
SOLVED:Ethanol (ethyl alcohol), CH3 CH2 OH, can act as a Brønsted-Lowry acid. Write the chemical equation for the reaction of ethanol as an acid with hydroxide ion, OH^-. Ethanol can also react